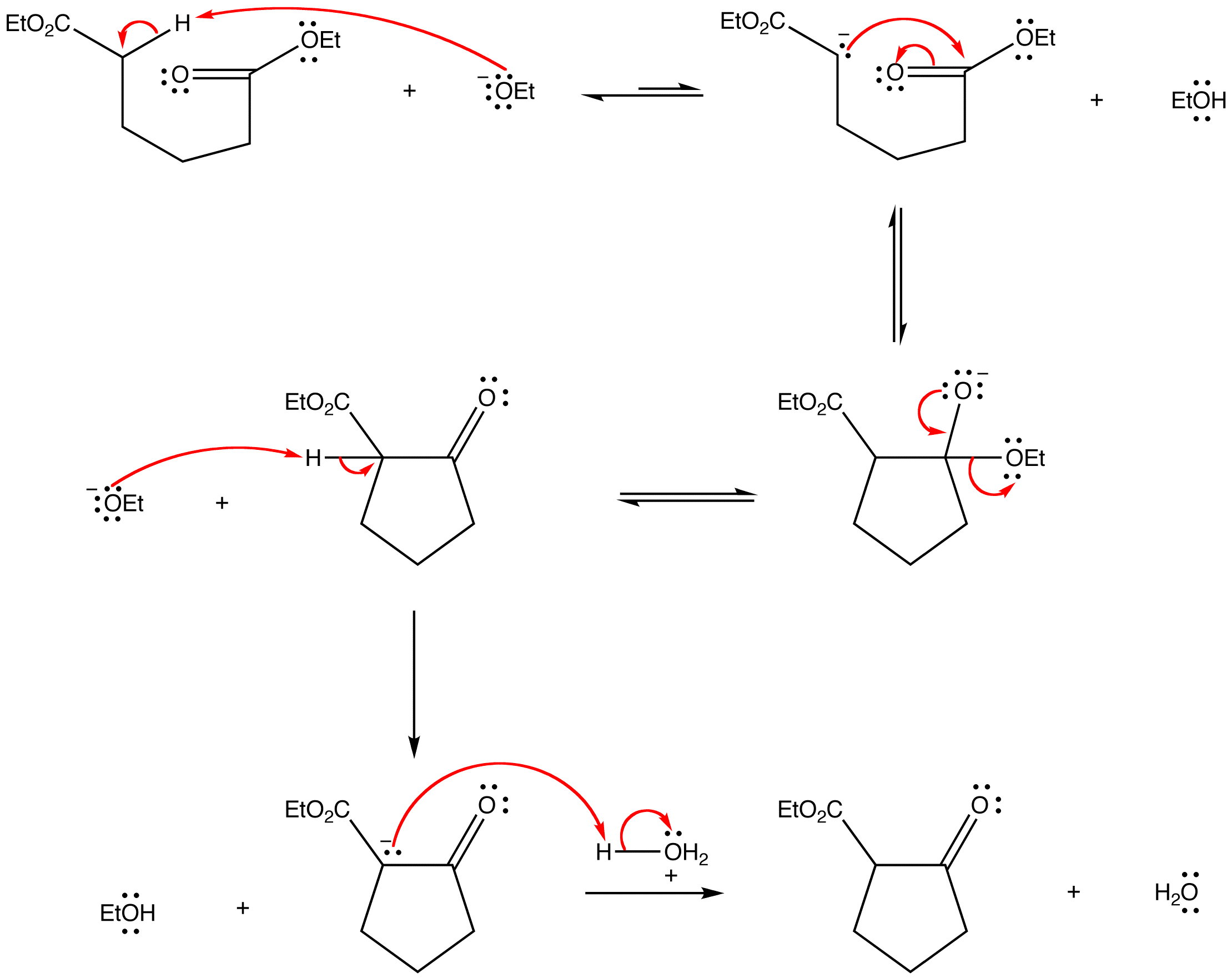
The Dieckmann condensation is the intramolecular chemical reaction of diesters with base to give β-keto esters. It is named after the German chemist Walter Dieckmann (1869–1925). The equivalent intermolecular reaction is the Claisen condensation. Dieckmann condensations are highly effective routes to 5-, 6-, and 7-member rings, but poor for larger rings.
Reaction mechanism
Deprotonation of an ester at the α-position generates an enolate ion which then undergoes a 5-exo-trig nucleophilic attack to give a cyclic enol. Protonation with a Brønsted–Lowry acid (H3O for example) re-forms the β-keto ester.
Due to the steric stability of five- and six-membered rings, these structures will preferentially be formed. 1,6 diesters will form five-membered cyclic β-keto esters, while 1,7 diesters will form six-membered β-keto esters.
Further reading
- Dieckmann, W. Ber. 1894, 27, 102 & 965
- Dieckmann, W. Ber. 1900, 33, 595 & 2670
- Dieckmann, W. Ann. 1901, 317, 51 & 93
See also
- Claisen condensation
- Gabriel–Colman rearrangement
- Thorpe reaction
References